Guidelines
Statement of the Polish Wound Management Association
on larval therapy in wound management
Beata Mrozikiewicz-Rakowska![]() 1, Marcin Tusiński2, Przemysław Lipiński
1, Marcin Tusiński2, Przemysław Lipiński![]() 3, Dariusz Bazaliński
3, Dariusz Bazaliński![]() 4,5, Jolanta Dynarska6, Lidia Czwakiel7, Aneta Zymon8, Bożena Mospan9, Katarzyna Malinowska10, Maciej Sopata
4,5, Jolanta Dynarska6, Lidia Czwakiel7, Aneta Zymon8, Bożena Mospan9, Katarzyna Malinowska10, Maciej Sopata![]() 11, Arkadiusz Jawień
11, Arkadiusz Jawień![]() 12,13
12,13
1 Department of Endocrinology, Medical Centre of Postgraduate Education, Warsaw, Poland
2 Celeris Medical Center, Krakow, Poland
3 Department of Wound Management, ARGO Medical Center, Lodz, Poland
4 Podkarpackie Specialist Oncology Centre. Specialist Hospital in Brzozow, Poland
5 Institute of Health Sciences, College of Medical Sciences, University of Rzeszow, Poland
6 Jolanta Dynarska Medical Center, Szczecin, Poland
7 St. Luke’s Provincial Hospital, SPZOZ in Tarnow, Poland
8 AZ MED Pain and Wound Treatment Centre, Krakow, Poland
9 Lubuskie Diabetes Centre, Zielona Gora, Poland
10 Fundation O!Rany, Medycyna Ran, Bydgoszcz, Poland
11 Department and Clinic of Palliative Medicine, Stationary Hospice of the Poznan University of Medical Sciences, Poland
12 Department of Vascular Surgery and Angiology, Collegium Medicum in Bydgoszcz, Nicolaus Copernicus University in Torun, Poland
13 Dr. A. Jurasz University Hospital No. 1 in Bydgoszcz, Poland
Correspondence address: Prof. Beata Mrozikiewicz-Rakowska, Department of Endocrinology, Medical Centre of Postgraduate Education, 99/103 Marymoncka St., 01-813 Warsaw, Warsaw, Poland, e-mail: klinendo@cmkp.edu.pl
Received: 1.08.2023
Accepted: 27.09.2023
Published: 30.09.2023
LECZENIE RAN 2023; 20 (3): 89–95
DOI: 10.60075/lr.v20i3.43
Table of contents:
Aim
Maggot debridement therapy (MDT) is a topical therapy for difficult-to-heal (chronic) wounds, recognised by scientific societies and healthcare system organisations in many countries for its contribution to the wound healing process [1, 2]. Maggot debridement therapy has been increasingly used in Poland for over a decade, both in outpatient and inpatient settings.
By issuing this document, the Polish Wound Management Association supports the use of this method by physicians and nurses licensed to treat wounds, subject to separate regulations. With a view to continually improving the quality of wound management in Poland, the Association recognises the need to standardise the principles of this therapy.
On account of the specific nature of MDT and wound treatment itself, the availability of scientific studies of the highest quality (i.e. randomised comparative studies) is very limited [3–7]. Consequently, this position statement relies on existing research findings including case reports, descriptions of case series, in vitro and basic science studies as well as the clinical experience of the authors.
The authors have identified several areas of dispute regarding the application of MDT, not addressed in this paper, with respect to which available scientific evidence does not allow formulating an unambiguous position. Also, in a number of contentious matters, where a clear scientific rationale was lacking, the authors have adopted a cautious stance.
Wound debridement using living Lucilia sericata larvae (MDT) is a safe and easy method to apply in clinical practice in both outpatient and inpatient care. Maggot debridement therapy is unique type of local therapy employing live insects in their larval stage [8, 9].
The key steps in preparing the wound bed include removing non-viable and necrotic tissue and reducing the bacterial burden. These measures help expedite the resolution of the inflammatory phase and promote an environment conducive to tissue healing and regeneration. The choice of the most appropriate wound debridement method should be based on factors including the origin of wound, wound condition, available resources, and the patient’s preferences [10–12].
Nomenclature
In the English-language literature, the terms maggot debridement therapy (MDT) and larval debridement therapy (LDT) are the most common [8, 13]. Throughout this paper, the term maggot debridement therapy (MDT) is used. Other names referring to this type of treatment include larval therapy, larva therapy, larvae therapy, biodebridement, and biosurgery.
Mechanism of action
Maggot debridement therapy has a multifaceted activity, producing mechanical, enzymatic, bactericidal and bacteriostatic effects, and promoting the healing process through direct physical contact of the larvae with the wound bed [13–15].
Immature larvae, which are unable to reproduce, are necrophagous. When residing within a wound, they feed actively and voraciously on necrotic tissue in the process of extracorporeal digestion. A single larva consumes approximately 25 mg of tissue over a 24-hour period [16].
The secretions produced by the larvae – known as defensins – are protein substances which possess bactericidal properties and promote wound healing processes within the wound bed. The strength of the proteolytic effects of the excretions and secretions, as well as their antibacterial, antibiofilm and anti-inflammatory activities and synergism with selected antibiotics, depend on the number of larvae in a specific area, type of microorganisms present, and the amount of exudate [13, 17–20].
Safety of therapy and medical staff qualification
Maggot debridement therapy is a safe method, carrying a low risk of adverse reactions, which are listed further down in the document.
Maggot debridement therapy may be carried out exclusively by licensed nurses and physicians. According to the regulations in place, nurses without a master’s degree are required to complete a course in wound care.
Types of maggot debridement therapy
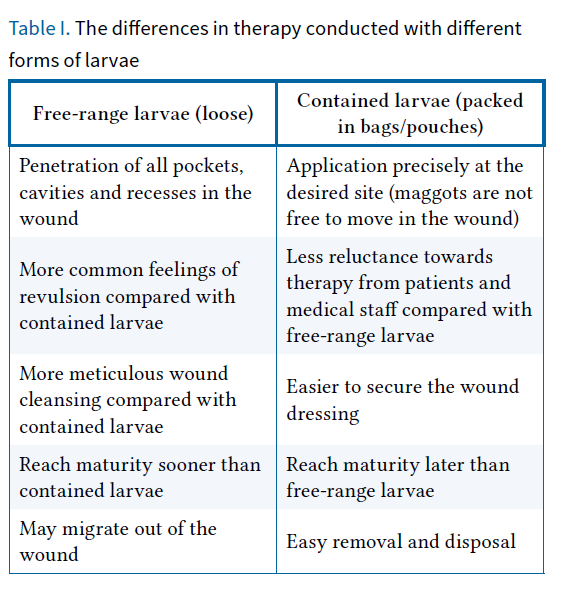
Larvae used for MDT are available in two forms: free-range and contained (bagged). The choice of therapy type is at the discretion of the treatment provider. The main differences in therapy conducted with different forms of larvae are outlined in the Table I [13, 14, 19, 21, 22].
Ordering, transport and storage
The manufacturer is responsible for the microbiological control and verification of the larval species [13, 23]. After receiving an order, the manufacturer prepares and delivers larvae according to the specifications (number and form of larvae) supplied by the treatment provider. For free-range larvae, the recommended dose is 5 to 10 larvae per square centimetre of wound surface area. Contained larvae are ordered by selecting the size and number of bags to suit the size and shape of the wound.
The manufacturer is responsible for supplying larvae that are ready for application, i.e. viable and at the appropriate stage of development. To this end, the manufacturer ensures a cold supply chain and uses appropriate packaging during transport from the laboratory to the medical facility or the location where the larvae will be applied to the patient. The manufacturer must guarantee appropriate transport conditions and duration so that the manufacturer’s expiry date is reached no earlier than 24 hours after the larvae are supplied to the designated
location.
The product packaging should allow batch identification and include information about the quantity of larvae, manufacturing date, and expiry date. The attached informational materials should clearly state that treatment can only be conducted by medical professionals licensed to provide wound care.
Larvae should be applied prior to the expiry date specified by the manufacturer; it is advisable to minimise the time between the delivery of larvae and their application. The storage of larvae during this period is the responsibility of the treatment provider. The recommended temperature range is 4–8oC.
Immediately prior to application, larval viability must be checked by visual assessment of motility. In cases of doubt about the viability of the larvae, the assessment can be repeated after warming the container to room temperature. Larvae must not be applied to the wound if any doubt remains as to whether they are viable.
Patient preparation [21, 22]
Informed written consent for the use of MDT must be obtained from the patient or legal guardian.
Before the application of larvae, the patient and caregiver should be provided with detailed information about:
- purpose and expected benefits of MDT,
- mechanism of action of MDT,
- procedure and conduct during therapy,
- course of therapy, including the recommended actions to be taken (e.g. changing the secondary dressing),
- potential discomfort associated with an unpleasant odour, seeing larvae migrate from the dressing, etc.,
- possible adverse reactions associated with therapy and ways to mitigate them,
- forms of communication with the treatment provider.
Preparation of wound and surrounding tissues [13, 14, 16, 21]
In wounds with a significant amount of necrotic tissue, we recommend pretreatment of the wound area by sharp debridement prior to the application of larvae.
Before the larvae are placed on the wound, it should be cleansed with a neutral solution, and loose fragments of dead tissue, exudate, and fibrin should be removed mechanically (Fig. 1A).
Because the substances secreted have irritant effects, it is advisable to protect the skin around the wound. Depending on wound type and location, and the individual characteristics of the patient’s skin, suitable skin protection products may include zinc-based ointments or pastes, stoma paste or hydrocolloid dressings (Fig. 1B).
Principles of larval application [13, 14, 21]
Free-range larvae
The two most commonly used methods of applying free-range larvae are described below:
1) using a pipette:
Add the saline solution to the vial, rinsing the larvae off the walls and stopper so that they sink to the bottom. Next, use a sterile disposable Pasteur pipette to draw up the fluid containing larvae and apply it into the wound (Fig. 2A). A single “thick” droplet contains 40 to 50 larvae. This type of application makes it easier to control the number of larvae placed in several wounds at the same time (Fig. 2B);
2) by rinsing:
Fold a sterile non-woven gauze pad into a boat shape. Add the saline solution to the vial, rinsing the larvae off the walls and stopper so that they sink to the bottom. Next, transfer the liquid slowly to the gauze pad, draining off the larvae. Repeat the rinsing process until all biological material has been collected on the gauze. Invert the gauze pad and place it over the wound so that the biological material is in direct contact with the wound.
After applying the larvae, cover the wound with a few layers of moist, sterile non-woven gauze. In the next step, apply gauze to an area larger than the wound and secure it with a bandage and plaster tape to prevent the dressing from slipping off during treatment. This is the primary wound dressing that should not be removed during MDT, unless there is an important reason to do so.
Place a secondary (absorbent) dressing made of gauze, or a commercially available superabsorbent dressing, on top and fix it to the primary dressing.
Contained larvae
Remove the bags (pouches) from the transport vial using a smooth-surfaced instrument to avoid perforation, and position them on the prepared wound bed (Fig. 1C). Secure the bags in place on the wound using a bandage to protect them from unintended shifting, but without applying excessive pressure (Fig. 1D, E). Fix the bandage with plaster tape. Place a secondary (absorbent) dressing made of gauze, or a commercially available superabsorbent dressing, on top and fix it to the primary dressing (Fig. 1F). Fasten the secondary dressing securely (Fig. 1G, H).
Patient management during maggot debridement therapy [13, 16, 21, 22]
The secondary dressing should be changed depending on the amount of exudate (usually 1 to 4 times a day) to minimize the risk of irritation in the wound area and improve the patient’s comfort by removing excess foul-smelling exudate.
When changing the secondary dressing, the primary dressing should be moistened with either water for injection or saline solution: always on the first day, and optionally also on subsequent days of therapy, depending on the amount of exudate, to maintain a moist environment.
Post-therapy management
Larvae should be removed on the third or fourth day after application. Remove the dressing and pull the dressing off the wound together with the free-range or contained larvae. Wash away any free larvae remaining in the wound with saline solution and remove them mechanically. Place all removed material in a medical waste bag. Apply isopropyl alcohol (a disinfectant) into the bag, as needed. Seal the bag tightly and dispose of according to the site’s medical waste disposal procedure.
After cleaning the wound and skin around the wound of any debris, initiate the follow-up treatment plan.
Maggot debridement therapy can be repeated until the desired clinical outcome is achieved.
Indications
Maggot debridement therapy is indicated in the treatment of wounds of known origin, without the contraindications listed below, which require debridement and preparation for further treatment modalites [1, 8, 9, 12, 14, 15, 23, 26].
The above indications include:
- wounds due to diabetic foot ulcers (of neuropathic, mixed and ischaemic origin), after prior revascularisation treatment,
Maggot debridement therapy in the management of diabetic foot ulcers reduces the duration of debridement and expedites the healing process [5, 20].
- lower leg ulcers (of arterial, venous, and mixed origin) after prior revascularisation treatment,
Maggot debridement therapy in the management of venous ulcers reduces the duration of debridement, but has no impact on the healing time [3, 4, 6, 7].
- pressure ulcers,
Maggot debridement therapy in the management of pressure ulcers reduces the duration of debridement, but data are lacking to determine whether it impacts the healing time [24].
- other wounds meeting the above criterion [13].
Maggot debridement therapy should be used as a technique of wound preparation for subsequent treatment stages, such as negative pressure therapy, intermediate-thickness skin grafting, or skin flap coverage.
Contraindications [8, 9, 13, 25, 27]
- Wound of undetermined origin.
- Suspected or confirmed autoimmune or autoinflammatory origin of wound.
- Presence of cancer/cancerous infiltration in the wound bed or margins.
- Wound of ischemic origin without prior successful revascularisation therapy.
- Anaemia requiring blood product transfusion.
- Dry necrosis in the wound.
- Immediate anatomical proximity to major vessels.
- Potential wound communication with body cavity (peritoneal cavity, pleural cavity, pericardium, mediastinum).
- Active bleeding in the wound area.
- Infection (including systemic infection) spreading beyond the wound bed.
- Fever.
- Uncontrolled pain associated with the presence of wound prior to the initiation of MDT.
- Adverse reactions during previous MDT.
- No possibility to relieve wound tension.
- Lack of adherence from patient and/or his/her legal guardians.
- No possibility to obtain informed consent from the patient or legal guardian.
- Pregnancy.
Relative contraindications
- Pressure at the site of larval application, associated with the risk of destruction of biological material.
- Previous spontaneous bleeding from wound.
- The benefit-to-risk ratio should be carefully evaluated on a case-by-case basis in patients with coagulation disorders and on antiplatelet and anticoagulant therapy [3].
- Maggot debridement therapy should not be used as the sole method of treating wound infections. For infections contained within the wound, MDT can be employed as an adjunct to conventional antimicrobial treatment [6, 18, 20].
- In patients treated with compression therapy, we recommend discontinuing it for the duration of MDT, unless there is a significant risk of wound deterioration. During the temporary break in compression therapy, elevation of the limb is the appropriate treatment [3, 4].
Adverse reactions
Maggot debridement therapy is associated with a very small number of severe adverse reactions.
The most common adverse reactionsinclude [7, 13, 16, 28]:
- irritation and maceration of surrounding skin,
- red skin,
- oedema in the wound area,
- pain,
- pruritus.
- Less common adverse reactions include:
- increased temperature or fever,
- minor bleeding,
- severe bleeding requiring management of blood vessel trapped in necrotic tissue.
Disclosure
The authors declare no conflict of interest.
References
- von Beckerath O, Kanya S, Gäbel G, et al. Use of maggot debridement therapy in hospitalised patients in Germany. Int Wound J 2020; 17: 10–15. DOI: 10.1111/iwj.13204.
- Probst S, Seppänen S, Gerber V, et al. EWMA document: home care – wound care. J Wound Care 2014; 23 (Suppl. 5a): S1–S44.
- Davies CE, Woolfrey G, Hogg N, et al. Maggots as a wound debridement agent for chronic venous leg ulcers under graduated compression bandages: A randomised controlled trial. Phlebology 2015; 30: 693–699. DOI: 10.1177/0268355514555386.
- Dumville JC, Worthy G, Bland JM, et al. Larval therapy for leg ulcers (VenUS II): randomized controlled trial. BMJ 2009; 338: b773. DOI: 10.1136/bmj.b773.
- Malekian A, Esmaeeli Djavid G, Akbarzadeh K, et al. Efficacy of Maggot Therapy on Staphylococcus aureus and Pseudomonas aeruginosa in Diabetic Foot Ulcers: A Randomized Controlled Trial. J Wound Ostomy Continence Nurs 2019; 46: 25–29. DOI: 10.1097/ WON.0000000000000496.
- Mudge E, Price P, Walkley N, Harding KG. A randomized controlled trial of larval therapy for the debridement of leg ulcers: results of a multicenter, randomized, controlled, open, observer blind, parallel group study [published correction appears in Wound Repair Regen 2014; 22: 290. Neal, Walkley [corrected to Walkley, Neal]]. Wound Repair Regen 2014; 22: 43–51. DOI: 10.1111/wrr.12127.
- Opletalová K, Blaizot X, Mourgeon B, et al. Maggot therapy for wound debridement: a randomized multicenter trial. Arch Dermatol 2012; 148: 432–438. DOI: 10.1001/archdermatol.2011.1895.
- Jafari A, Hosseini SV, Hemmat HJ, Khazraei H. Lucillia Sericata larval therapy in the treatment of diabetic chronic wounds. J Diabetes Metab Disord 2022; 21: 305–312. DOI: 10.1007/s40200-022-00973-w.
- Moya-López J, Costela-Ruiz V, García-Recio E, et al. Advantages of Maggot Debridement Therapy for Chronic Wounds: A Bibliographic Review. Adv Skin Wound Care 2020; 33: 515–525. DOI: 10.1097/01. ASW.0000695776.26946.68.
- Sibbald RG, Elliott JA, Persaud-Jaimangal R, et al. Wound bed preparation 2021. Adv Skin Wound Care 2021; 34: 183–195.
- Atkin L, Bućko Z, Conde Montero E, et al. Implementing TIMERS: the race against hard-to-heal wounds. J Wound Care 2019; 28 (3 Suppl. 3): 1-49.L.
- Skórka M, Bazaliński D, Gajdek M, et al. Debridement of hard-to-heel wounds provided in the home-care setting. Practical and legal possibilities. Leczenie Ran 2022; 19: 84–93. DOI: 10.5114/lr.2022.120199.
- Sherman RA. Mechanisms of maggot-induced wound healing: what do we know, and where do we go from here? Evid Based. Complement Alternat Med 2014; 2014: 592419.
- Sherman RA, Shimoda KJ. Presurgical maggot debridement of soft tissue wounds is associated with decreased rates of postoperative infection. Clin Infect Dis 2004; 39: 1067–1070. DOI: 10.1086/423806.
- Elraiyah T, Domecq JP, Prutsky G, et al. A systematic review and meta-analysis of débridement methods for chronic diabetic foot ulcers. J Vasc Surg 2016; 63 (2 Suppl): 37S–45S.e2. DOI: 10.1016/j.jvs.2015.10.002.
- Bazaliński D, Kózka M, Karnas M, Więch P. Effectiveness of Chronic Wound Debridement with the Use of Larvae of Lucilia Sericata. J Clin Med 2019; 8: 1845. DOI: 10.3390/jcm8111845.
- Tombulturk FK, Kanigur-Sultuybek G. A molecular approach to maggot debridement therapy with Lucilia sericata and its excretions/secretions in wound healing. Wound Repair Regen 2021; 29: 1051–1061. DOI: 10.1111/wrr.12961.
- Gazi U, Taylan-Ozkan A, Mumcuoglu KY. The effect of Lucilia sericata larval excretion/secretion (ES) products on cellular responses in wound healing. Med Vet Entomol 2021; 35: 257–266. DOI: 10.1111/mve.12497.
- Bazaliński D, Skórka M, Szymańska P, et al. Lucilia sericata defensins and their capacity to stimulate wound regenerative processes. A review of the literature and our own observations. Leczenie Ran 2022; 19: 94–105. DOI: 10.5114/lr.2022.120200.
- Sun X, Chen J, Zhan J, et al. Maggot debridement therapy promotes diabetic foot wound healing by up-regulating endothelial cell activity. J Diabetes Complications 2016; 30: 318–322
- Szewczyk M, Cwajda-Białasik J, Mościcka P, et al. Treatment of pressure ulcers – recommendations of the Polish Wound Management Association. Part II. Leczenie Ran. 2020; 18: 151–184. DOI: 10.5114/lr.2020.103116.
- Bazaliński D. Skuteczność terapii biologicznej z wykorzystaniem larw Lucilia sericata w leczeniu ran przewlekłych u chorych w opiece długoterminowej i paliatywnej. Wydawnictwo Uniwersytetu Rzeszowskiego, Rzeszów 2019.
- Zarchi K, Jemec GBE. The efficacy of maggot debridement therapy a review of comparative clinical trials. Int Wound J 2012; 9: 469–477.
- Sherman RA. Maggot versus conservative debridement therapy for the treatment of pressure ulcers. Wound Repair Regen 2002; 10: 208–214. DOI: 10.1046/j.1524-475x.2002.10403
- Morozov AM, Sherman RA. Survey of patients of the Tver region of Russia regarding maggots and maggot therapy. Int Wound J 2019; 16: 401–405.
- Shi E, Shofler D. Maggot debridement therapy: a systematic review. Br J Community Nurs 2014; Suppl Wound Care: S6–13.
- Tian X, Liang XM, Song GM, et al. Maggot debridement therapy for the treatment of diabetic foot ulcers: a meta-analysis. J Wound Care 2013; 22: 462–469. DOI: 10.12968/jowc.2013.22.9.462.
- Mumcuoglu KY, Davidson E, Avidan A, Gilead L. Pain related to maggot debridement therapy. J Wound Care 2012; 21: 400, 402, 404–405.


2 thoughts on “LECZENIE RAN 2023; 20 (3)”