Original paper
Polish honeys exhibit activity against Pseudomonas aeruginosa isolated from wounds
Tomasz M. Karpiński![]() 1, Joanna Karpińska
1, Joanna Karpińska![]() 2, Marcin Ożarowski
2, Marcin Ożarowski![]() 3
3
1 Chair and Department of Medical Microbiology, Poznan University of Medical Sciences, Poznan, Poland
2 Antimicrobials Research Group (ARG), Universal Scientific Education and Research Network (USERN), Poland
3 Department of Biotechnology, Institute of Natural Fibres and Medicinal Plants – National Research Institute, Poznan, Poland
Address for correspondence
Tomasz M. Karpiński, Chair and Department of Medical Microbiology, Poznan University of Medical Sciences, 10 Rokietnicka St., 60-806 Poznan, Poland,, e-mail: tkarpin@ump.edu.pl
Table of contents:
Abstract
Pseudomonas aeruginosa is recognised as an opportunistic pathogen and a common contributor to wound infections. This study focused on evaluating the antibacterial activity of selected Polish honeys against P. aeruginosa. Six bacterial strains isolated from wounds and 4 types of honeys (multifloral, linden, buckwheat, and heather) were utilised. The minimal inhibitory concentrations (MIC) were determined through the micro-dilution method employing 96-well plates, while antibiofilm activity was assessed using fluorescence microscopy after a 4-hour incubation period with the honeys. The results indicated diverse antibacterial efficacy among honeys, with linden and heather honeys demonstrating the most potent activity. Linden honey displayed an average MIC of 20.8%, while heather honey exhibited an MIC of 25%. Both honeys significantly reduced biofilm formation, with linden honey achieving a reduction of 54–67% and heather honey of 45-58%. Buckwheat and multifloral honeys exhibited lower activity, demonstrating average MIC levels of 37.5% and 41.7%, respectively, and a biofilm reduction below 20%. Conclusively, Polish honeys exhibited promising activity against both planktonic and biofilm forms of P. aeruginosa. These findings suggest potential applications in wound healing treatments, emphasising the need for further research in this area. The study contributes valuable insights to the limited literature on the antibacterial activity of Polish honeys.
Key words: bee product, antimicrobial, treatment.
Introdution
Pseudomonas aeruginosa is an aerobic Gram-negative bacillus, recognised as an opportunistic pathogen and a primary contributor to wound infections. In chronic wounds, the prevalence of P. aeruginosa varies from a few to approximately 40% [1]. However, studies by de Oliveira et al. [2] have demonstrated that this prevalence can escalate to as high as 75%. The bacterium is equipped with flagella, pili, and fimbriae. It produces an array of virulence factors, including pigments, elastase, phospholipase, protein convertase, enterotoxin, and exotoxin A. These virulence factors play a role in the degradation of skin and mucous membrane structures [3, 4]. Furthermore, research indicates that 100% of P. aeruginosa strains have the capability of forming biofilms [3], impeding the effectiveness of antibiotics and antiseptics. Together with Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, and Enterobacter spp., P. aeruginosa is classified as a multi-resistant pathogen under the acronym ESKAPE [5]. Carbapenem-resistant P. aeruginosa is designated by the WHO as a “critical” pathogen urgently necessitating new antibiotics [6]. Pseudomonas aeruginosa exhibits intrinsic resistance to several antibiotics, including sulphonamides, ampicillin, first- and second-generation cephalosporins, chloramphenicol, and tetracycline [7]. Acquired resistance, affecting over 10% of P. aeruginosa strains, extends to antibiotics such as aztreonam, ciprofloxacin, imipenem, meropenem, ceftazidime, cefotaxime, gentamicin, and piperacillin [2, 3]. Recently, strains exhibiting adaptation or resistance to antiseptics such as triclosan, chlorhexidine, benzalkonium chloride, and polyhexamethylene biguanide (PHMB) have also been identified [8, 9].
In line with Polish guidelines [10] and international consensus [11], wound treatment should involve the application of lavaseptics and antiseptics based on octenidine, polyhexanide, iodine-based povidone, or chlorhexidine. Nevertheless, the scientific community continues to explore the antimicrobial properties of natural substances or novel chemical compounds. Numerous studies highlight the potent antimicrobial effects of honey, particularly Manuka honey [12–14]. However, limited research exists regarding the antibacterial activity of Polish honeys [15, 16]. Therefore, the objective of this study was to investigate the efficacy of selected Polish honeys against P. aeruginosa and its biofilm.
Material and methods
Pseudomonas aeruginosa strains
The experiments were conducted using 6 strains of P. aeruginosa isolated from wounds and are part of the collection at the Department of Medical Microbiology, PUMS. All strains were cultivated at 37°C for 24 hours on Cetrimide agar (Graso Biotech, Poland).
Honeys
Four honeys were used in the research: multifloral, linden, buckwheat and heather. All these honeys originated from the Greater Poland Voivodeship, Poland. The honeys were directly dissolved in Mueller Hinton broth (Graso Biotech, Poland).
Minimal Inhibitory Concentrations
The minimal inhibitory concentrations (MIC) of the honeys were determined using the micro-dilution method with 96-well plates (Nest Scientific Biotechnology, China). The experimental procedures followed the methodology outlined in our previous publications [17, 18]. In summary, each well received 90 µl of Mueller Hinton broth (Graso Biotech, Poland) and 10 µl of bacterial suspension, resulting in a final inoculum concentration of 105 CFU/ml, as per McFarland standards. Serial dilutions of honey were carried out during the course of the study. The plates were then incubated at 37°C for 24 hours, and MIC values were determined through visual analysis.
Anti-biofilm activity test
Biofilm formation occurred in 12-well plates with Mueller Hinton broth over 48 hours at 37°C. After incubation, the wells were rinsed with PBS, and 1 mL of 50% honey water solution was introduced for 4 hours. Following this, the plates underwent three PBS washes. The assessment of anti-biofilm activity was conducted using the LIVE/DEAD BacLight Bacterial Viability Kit (Invitrogen, USA), following the manufacturer’s instructions. The results were observed using a fluorescence microscope Leica DM 1000 LED fluorescence microscope (Leica, Germany) with a Progres Gryphax camera (Jenoptik, Germany). The acquired images were analysed using Fiji, a distribution of the image analysis program ImageJ [19]. The program was employed to calculate the number of bacterial cells within the biofilm following the action of the honeys.
Results
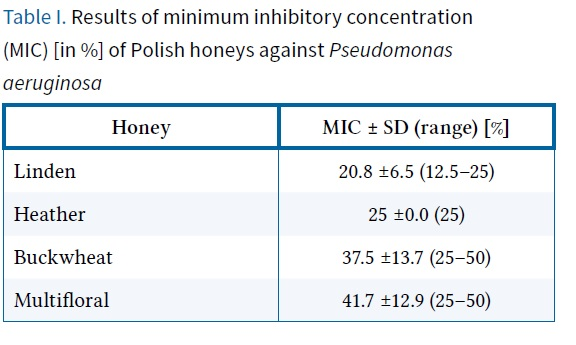
the research demonstrated varied antibacterial activity among the honeys. In terms of minimum inhibitory concentration (MIC), the most effective activity was exhibited by linden and heather honey, with MICs at average levels of 20.8% and 25%, respectively (Table I). Buckwheat and multifloral honeys showed weaker activity, with average MIC levels of 37.5% and 41.7%, respectively.
Similarly to MIC results, superior effectiveness of linden and heather honeys was observed in the assessment of antibiofilm activity. Linden honey led to a reduction in biofilm by 54–67% after 4 hours of exposure, while heather honey showed a reduction at the level of 45–58%. Buckwheat and multifloral honeys exhibited much weaker antibiofilm effects, with their biofilm reduction below 20% (Figure 1).
Discussion
There is limited literature on the antimicrobial activity of Polish honeys. Therefore, we compared the obtained results with those of European honeys. The activity of linden honey from Hungary was diverse, with MIC values against P. aeruginosa ranging from 12.5 to 50% [20, 21]. Linden honey from the Western Balkans exhibited activity against Gram-negative bacteria with MIC values of 12.5–25% [22]. Linden and heather honeys from Ukraine exhibited similar activities (MICs 19–38%) as those observed in the presented studies [23]. The mean MIC of Portuguese heather honey ranged from 10 to 13.3 for P. aeruginosa, indicating slightly better activity compared to ours [24]. In the case of multifloral honeys, other studies indicate better antibacterial activity. The MICs of honeys from Switzerland against P. aeruginosa were around 10–25% [25], and for honeys from Ukraine against Gram-negative bacteria, it ranged from 19 to 38% [23]. The MIC values for multifloral honeys from Poland ranged from 3.1% to > 12.5%, but this activity was observed against Staphylococcus aureus [26]. Moreover, in this study, MIC values > 12.5% were not analysed, meaning that for some honeys, this value could have been much higher. In the same paper, the MIC values against S. aureus for buckwheat honeys from Poland ranged from 1.56% to > 12.5%, but higher levels were also not analysed [26].
There are few papers on the antibiofilm activity of European honeys. Inhibitory rates of Hungarian linden honey were approximately 60 to 85% [20, 21]. The difference compared to our studies may result from the fact that in the Hungarian research, biofilm formation was conducted for only 4 hours, meaning it was not mature and could be more susceptible to the compounds present in honey. In antibiofilm studies of Portuguese heather honey [27], a different methodology was applied, examining the logarithmic reduction in colony count. A 50% honey concentration led to approximately 50% decrease in the logarithm of colony count.
Currently, most publications focus on Manuka honey, and bandages and gels containing this honey are available in the market. However, the MIC values for Manuka honeys against P. aeruginosa range from 6% to 33% [12, 28–31]. This implies that Polish honeys, such as linden and heather, do not lag behind Manuka honey in terms of antibacterial activity. Therefore, it is worthwhile for Polish honeys to also find application in wound treatment.
Conclusions
The presented studies indicate that Polish honeys are active against both planktonic and biofilm forms of Pseudomonas aeruginosa. Further research is needed to explore their potential use in wound healing treatments.
Acknowledgments
We would like to thank the Beekeeping Farm “Miody Bartkowiaka” (Konarzewo, Poland), for their support in the research.
Disclosure
The authors declare no conflict of interest.
This research received no external funding.
Approval of the Bioethics Committee is not required.
References
- Phan S, Feng CH, Huang R, et al. Relative Abundance and Detection of Pseudomonas aeruginosa from Chronic Wound Infections Globally. Microorganisms 2023; 11: 1210. DOI: https://doi.org/10.3390/microorganisms11051210.
- de Oliveira FP, Pires BMFB, de Cássia Ferreira de Almeida Silva K, et al. Prevalence, Antimicrobial Susceptibility, and Clonal Diversity of Pseudomonas aeruginosa in Chronic Wounds. J Wound Ostomy Continence Nurs 2017; 44: 528–535. DOI: https://doi.org/10.1097/WON.0000000000000373.
- Ghasemian S, Karami-Zarandi M, Heidari H, et al. Molecular characterizations of antibiotic resistance, biofilm formation, and virulence determinants of Pseudomonas aeruginosa isolated from burn wound infection. J Clin Lab Anal 2023; 37: e24850. DOI: https://doi.org/10.1002/jcla.24850.
- Qin S, Xiao W, Zhou C, et al. Pseudomonas aeruginosa: pathogenesis, virulence factors, antibiotic resistance, interaction with host, technology advances and emerging therapeutics. Sig Transduct Target Ther 2022; 7: 1–27. DOI: https://doi.org/10.1038/s41392-022-01056-1.
- Gauba A, Rahman KM. Evaluation of Antibiotic Resistance Mechanisms in Gram-Negative Bacteria. Antibiotics (Basel) 2023; 12: 1590. DOI: https://doi.org/10.3390/antibiotics12111590.
- WHO publishes list of bacteria for which new antibiotics are urgently needed n.d. Available at: https://www.who.int/news/item/27-02-2017-who-publishes-list-of-bacteria-for-which-new-antibiotics-are-urgently-needed (Access: December 27, 2023).
- Reygaert WC. An overview of the antimicrobial resistance mechanisms of bacteria. AIMS Microbiol 2018; 4: 482–501. DOI: https://doi.org/10.3934/microbiol.2018.3.482.
- Rozman U, Pušnik M, Kmetec S, et al. Reduced Susceptibility and Increased Resistance of Bacteria against Disinfectants: A Systematic Review. Microorganisms 2021; 9: 2550. DOI: https://doi.org/10.3390/microorganisms9122550.
- Monteiro Vasconcelos F, Cabral Pereira da Costa C, Peres EM, et al. Microbiological identification and resistance profile of microorganisms in pressure injuries after the use of polyhexamethylene biguanide: a series of fourteen cases. Wounds 2022; 33: 51–56. DOI: https://doi.org/10.25270/wnds/2022.5156.
- Sopata M, Jawień A, Mrozikiewicz-Rakowska B, et al. Guidelines for topical management in non-infected, at risk of infection and infected wounds – an overview of available antimicrobial substances used in the treatment of wounds. Recommendations of the Polish Wound Treatment Society [in Polish]. Leczenie Ran 2020; 17: 1–21. DOI: https://doi.org/10.5114/lr.2020.96820.
- Nair HKR, Mrozikiewicz-Rakowska B, Pinto DS, et al. International Consensus Document: Use of wound antiseptics in practice. Wounds International 2023; 1–28.
- Camplin AL, Maddocks SE. Manuka honey treatment of biofilms of Pseudomonas aeruginosa results in the emergence of isolates with increased honey resistance. Ann Clin Microbiol Antimicrob 2014; 13: 19. DOI: https://doi.org/10.1186/1476-0711-13-19.
- Hewett SR, Crabtrey SD, Dodson EE, et al. Both Manuka and Non-Manuka Honey Types Inhibit Antibiotic Resistant Wound-Infecting Bacteria. Antibiotics (Basel) 2022; 11: 1132. DOI: https://doi.org/10.3390/antibiotics11081132.
- Mokhtar JA, McBain AJ, Ledder RG, et al. Exposure to a Manuka Honey Wound Gel Is Associated With Changes in Bacterial Virulence and Antimicrobial Susceptibility. Front Microbiol 2020; 11: 2036. DOI: https://doi.org/10.3389/fmicb.2020.02036.
- Gośliński M, Nowak D, Kłębukowska L. Antioxidant properties and antimicrobial activity of manuka honey versus Polish honeys. J Food Sci Technol 2020; 57: 1269–1277. DOI: https://doi.org/10.1007/s13197-019-04159-w.
- Kačániová M, Borotová P, Galovičová L, et al. Antimicrobial and Antioxidant Activity of Different Honey Samples from Beekeepers and Commercial Producers. Antibiotics (Basel) 2022; 11: 1163. DOI: https://doi.org/10.3390/antibiotics11091163.
- Adamczak A, Ożarowski M, Karpiński TM. Curcumin, a Natural Antimicrobial Agent with Strain-Specific Activity. Pharmaceuticals (Basel) 2020; 13: 153. DOI: https://doi.org/10.3390/ph13070153.
- Karpiński TM, Ożarowski M, Seremak-Mrozikiewicz A, Wolski H. Anti-Candida and Antibiofilm Activity of Selected Lamiaceae Essential Oils. Front Biosci (Landmark Ed) 2023; 28: 28. DOI: https://doi.org/10.31083/j.fbl2802028.
- Fiji. ImageJ Wiki n.d. Available at: https://imagej.github.io/software/fiji/index (Access January 1, 2024).
- Balázs VL, Nagy-Radványi L, Bencsik-Kerekes E, et al. Antibacterial and Antibiofilm Effect of Unifloral Honeys against Bacteria Isolated from Chronic Wound Infections. Microorganisms 2023; 11: 509. DOI: https://doi.org/10.3390/microorganisms11020509.
- Farkas Á, Balázs VL, Kõszegi T, et al. Antibacterial and Biofilm Degradation Effects of Hungarian Honeys Linked With Botanical Origin, Antioxidant Capacity and Mineral Content. Front Nutr 2022; 9: 953470. DOI: https://doi.org/10.3389/fnut.
2022.953470. - Sakač M, Jovanov P, Marić A, et al. Antioxidative, Antibacterial and Antiproliferative Properties of Honey Types from the Western Balkans. Antioxidants (Basel) 2022; 11: 1120. DOI: https://doi.org/10.3390/antiox11061120.
- Cilia G, Fratini F, Marchi M, et al. Antibacterial Activity of Honey Samples from Ukraine. Vet Sci 2020; 7: 181. DOI: https://doi.org/10.3390/vetsci7040181.
- Feás X, Iglesias A, Rodrigues S, Estevinho LM. Effect of Erica sp. honey against microorganisms of clinical importance: study of the factors underlying this biological activity. Molecules 2013; 18: 4233–4246. DOI: https://doi.org/10.3390/molecules18044233.
- Godocikova J, Bugarova V, Kast C, et al. Antibacterial potential of Swiss honeys and characterisation of their bee-derived bioactive compounds. J Sci Food Agric 2020; 100: 335–342. DOI: https://doi.org/10.1002/jsfa.10043.
- Grecka K, Kuś PM, Worobo RW, Szweda P. Study of the Anti-Staphylococcal Potential of Honeys Produced in Northern Poland. Molecules 2018; 23: 260. DOI: https://doi.org/10.3390/molecules23020260.
- Fernandes L, Oliveira A, Henriques M, Rodrigues ME. Honey as a Strategy to Fight Candida tropicalis in Mixed-Biofilms with Pseudomonas aeruginosa. Antibiotics (Basel) 2020; 9: 43. DOI: https://doi.org/10.3390/antibiotics9020043.
- Anthimidou E, Mossialos D. Antibacterial activity of Greek and Cypriot honeys against Staphylococcus aureus and Pseudomonas aeruginosa in comparison to manuka honey. J Med Food 2013; 16: 42–47. DOI: https://doi.org/10.1089/jmf.2012.0042.
- Bucekova M, Godocikova J, Gueyte R, et al. Characterisation of physicochemical parameters and antibacterial properties of New Caledonian honeys. PLoS One 2023; 18: e0293730. DOI: https://doi.org/10.1371/journal.pone.0293730.
- Al-Kafaween MA, Nagi Al-Jamal HA. A comparative study of antibacterial and antivirulence activities of four selected honeys to Manuka honey. Iran J Microbiol 2022; 14: 238–251. DOI: https://doi.org/10.18502/ijm.v14i2.9193.
- Girma A, Seo W, She RC. Antibacterial activity of varying UMF-graded Manuka honeys. PLoS One 2019; 14: e0224495. DOI: https://doi.org/10.1371/journal.pone.0224495.

